Abstract:
Purpose
Extensive surgical procedure or liver transplant still remains the gold standard for treating slow-growing tumors in liver. But only few candidates are suitable for such procedure due to poor liver function, tumors in unresectable locations or presence of other liver diseases. In such situations minimally invasive surgery may be the best therapeutic procedure. The success of minimally invasive procedure depends on accurately targeting the desired region and guiding the entire procedure. The purpose of this study is to use a combined ultrasound imaging and electromagnetic tracking system to accurately place a steerable acoustic needle ablator and multiple temperature sensors in pig liver in vivo.
Methods
Catheter based interstitial ultrasound applicator of center frequency of 6-7 MHz was used to ablate in vivo pig liver under gas anesthesia. Temperature sensors were placed at eight different locations to estimate thermal distribution in the three-dimensional treated volume. The ultrasound imaging was acquired using a SonixTouch (Ultrasonix, Richmond, BC, Canada) system with an L14-5/38 GPS probe. The in-house software architecture and interface was developed to communicate with the hardware, ultrasound imaging and tracking system.
Results
During therapy a maximum temperature of 60-70 °C and dose of 104-106 equivalent minutes at 43°C was observed at a radial distance 15-20 mm from the center of the ablation transducer for a treatment time of 7-9 minutes. The dose distribution was analyzed and compared with the gross pathology (Fig.1) of the treated region. Accurate placement of the acoustic applicator and temperature sensors were achieved using the combined imageguidance and the tracking system. Note control of energy direction to avoid other structures.
Conclusions
The experimental results demonstrated that the directionality and shape of the ablation zone can be controlled using catheter based high intensity sectored ultrasound transducers. The 180° sectored transducers helped in creating desired ablation pattern without damaging the nearby vein/vessels in the tissue verified from gross pathology inspection (Fig. 1(a)). Our results suggest that tracked targeting coupled with directionality and shape of the ablated region could accurately control ablation zones using the proposed technology.
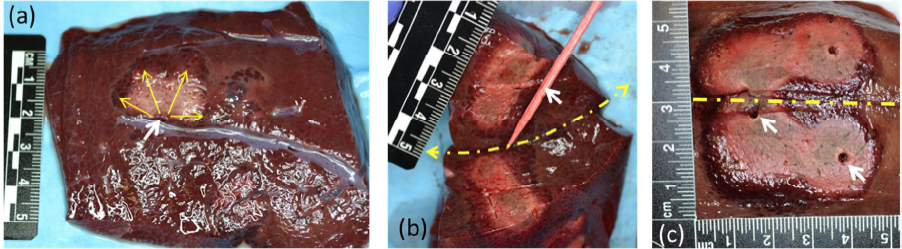
Figure 1: Gross pathology of the ablated pig liver tissue with (a) 180° radiation pattern single needle applicator (yellow solid arrows indicate the beam pattern) avoiding and (b) depth of abated region by 180° pattern applicator for tissue cut along long axis of needle (applicator location indicated by wooden stick), (c) pattern of 360° radiation pattern applicator (white arrows indicate the applicator position, dashed yellow arrow indicates the line of cut for tissue dissection).
